Small metal parts fail silently. A bolt rusts, a fastener seizes, a stamping corrodes — and a production line stops. Zinc barrel plating exists to prevent exactly this. This guide explains the science behind how zinc stops corrosion, walks through the complete barrel plating process step by step, and shows why it remains the most cost-effective corrosion protection available for high-volume small components.
If you manufacture small steel components at scale, corrosion isn’t a hypothetical risk — it’s an engineering problem that demands a real solution. A single corroded fastener in an automotive assembly, an agricultural machine, or a piece of industrial equipment can trigger failures that cost far more than the part itself. Zinc barrel electroplating exists specifically to prevent this outcome, and it does so with an efficiency and economy that no other coating process can match for small, high-volume parts.
But how does zinc actually stop corrosion? And why does the barrel plating process deliver such consistent, reliable protection across thousands of parts simultaneously? This guide answers both questions — and everything in between.
The Science: How Zinc Stops Corrosion
To understand why zinc barrel plating works, you first need to understand what causes corrosion in the first place — and why zinc is uniquely suited to stop it.
Steel corrodes through a process called oxidation. When iron atoms in steel are exposed to oxygen and moisture, they undergo an electrochemical reaction that forms iron oxide — what we commonly call rust. Once red rust appears on a steel part, the degradation accelerates, structural integrity diminishes, and the component’s service life is dramatically shortened.
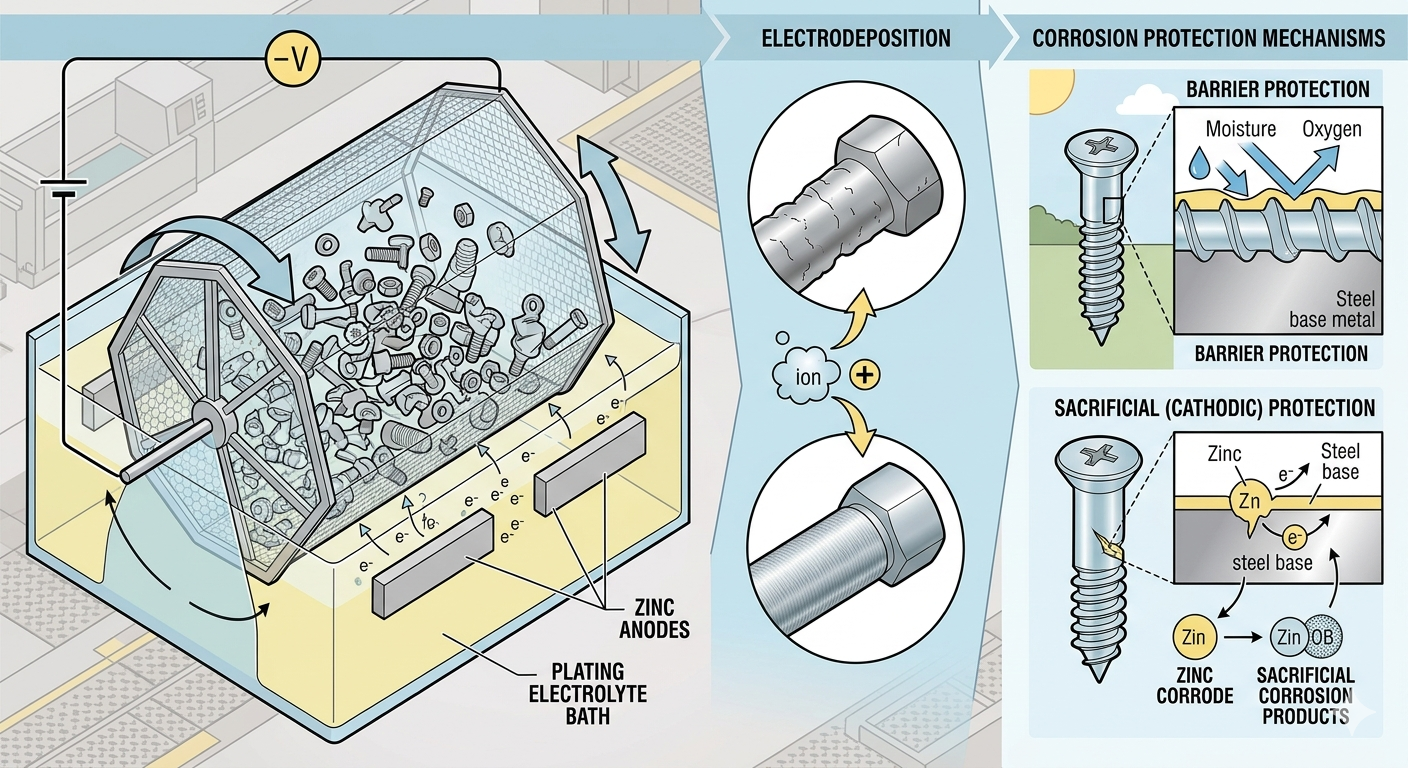
Zinc stops this process through two distinct mechanisms that work in combination: barrier protection and sacrificial (galvanic) protection.
Barrier Protection
The zinc coating applied during barrel plating forms a continuous physical barrier between the steel substrate and the corrosive environment. Oxygen, moisture, road salt, agricultural chemicals, and industrial contaminants cannot reach the underlying steel as long as the zinc layer remains intact. This is the first line of defense — and in many controlled environments, it’s sufficient on its own.
Sacrificial (Galvanic) Protection
This is where zinc’s chemistry becomes genuinely remarkable. Zinc sits lower than steel on the galvanic series — meaning when zinc and steel are in contact in the presence of an electrolyte (such as moisture), zinc corrodes preferentially. It sacrifices itself to protect the base steel.
In practical terms: even if a zinc-coated part is scratched, nicked, or has an area of bare steel exposed, the surrounding zinc will continue protecting that exposed area through galvanic action — a property called cathodic protection. The zinc corrodes first, appearing as white corrosion (zinc oxide), while the steel underneath remains unaffected. Only when the zinc is fully depleted does red corrosion (iron rust) begin to form.
Layer 3 — Passivate / chromate conversion coating Seals & extends protection
Layer 2 — Zinc deposit (sacrificial layer) Corrodes before steel
Layer 1 — Steel substrate (base metal) Protected underneath
This two-layer defense system — barrier plus sacrificial protection — is why zinc barrel plating consistently outperforms simple paint coatings or surface sealants for steel components exposed to harsh environments. Paint can be scratched through to bare steel with no remaining protection. Zinc keeps working even after the coating surface is damaged.
800 lbs
Capacity per barrel load at Plateco
0.072″
Smallest part diameter processable
ASTM B633
Primary industry spec for zinc on steel
Why Barrel Plating Is the Right Process for Small Parts
Zinc plating can be applied through several different processes — rack plating, mechanical galvanizing, and barrel plating. For small parts produced in high volumes, barrel plating is almost always the correct choice, and the reason comes down to how the process handles both geometry and scale.
In barrel plating, thousands of small components are loaded together into a perforated rotating barrel submerged in a zinc electrolyte solution. As the barrel tumbles, parts continuously reorient relative to the electrical current and plating chemistry. This continuous motion solves a fundamental problem in metal finishing: uniform coverage on complex geometries.
Threads, blind holes, recessed features, undercuts — these are exactly the areas most vulnerable to corrosion, and exactly the areas most difficult to plate using stationary methods. The tumbling motion of barrel plating naturally exposes every surface to the plating solution from multiple angles, ensuring consistent zinc deposition even in the hardest-to-reach features. Blind holes, which can trap air pockets in other processes, drain and fill naturally as the barrel rotates — eliminating the air pocket problem entirely.
“Barrel electroplating is also perfect for those smaller parts with blind holes as the rotation of the barrel allows the holes to fill up with solution as well as properly drain, meaning there are no issues with air pockets or bleed out.”
— Plateco, Inc., Zinc Barrel Electroplating
The electrical contact mechanism in barrel plating is also worth understanding. Because parts are tumbling and contacting each other, electricity transfers through part-on-part contact — similar to how touching a charged object transfers charge through physical contact. This distributed contact ensures that even parts not directly touching an electrical conductor still receive proper zinc deposition through contact with neighboring parts in the barrel.
The Zinc Barrel Plating Process — Step by Step
Effective corrosion protection doesn’t happen by accident. Every barrel plating run follows a precise sequence of steps, each critical to the quality of the final coating. Here is what the complete process looks like from part loading to finished certificate.
1 Bulk part loading
Large quantities of small steel parts are loaded into a perforated rotating barrel. The perforated design allows plating solution to flow freely through the barrel while maintaining electrical contact between parts as they tumble. Automated lines like those at Plateco can process up to 800 pounds per barrel load — one of the highest throughput capacities in the industry.
2 Cleaning and surface preparation
This is arguably the most critical step in the entire process. Parts arrive from manufacturing with oils, lubricants, heat treat residues, welding flux, and surface oxides — all of which will prevent zinc from bonding properly to the steel if not removed. The cleaning sequence involves degreasing, acid pickling, and thorough rinsing. At Plateco, over half of all processing tanks are dedicated entirely to cleaning — a testament to how foundational this step is to final coating quality. Cleaners are integrated directly into the production lines, eliminating the need for pre-cleaning and reducing lead times.
3 Zinc deposition in the electrolyte bath
Once cleaned, the barrel is lowered into a zinc electrolyte solution. Electrical current passes through the solution, driving zinc ions to migrate and deposit onto the negatively charged steel surfaces. The continuous tumbling motion ensures every surface — including threads, blind holes, and recessed features — gets consistent exposure to the solution, building an even zinc layer across the entire part. Chemistry balance, current density, temperature, and bath contamination are monitored throughout by automated systems to ensure repeatability from one run to the next.
4 Chromate conversion coating (passivation)
After zinc deposition, parts receive a passivate — a chromate conversion coating that dramatically boosts corrosion resistance and determines the part’s final appearance. Passivate options include trivalent clear (blue), trivalent yellow, trivalent black, hexavalent black, and olive drab, plus various dyed colors. Yellow passivate generally provides greater corrosion resistance than clear passivate due to the thicker conversion layer it forms. This post-treatment can push salt spray performance well beyond what bare zinc alone achieves.
5 Quality inspection and final rinse
Finished parts are inspected for coating thickness, appearance, and adherence to customer or industry specifications. At Plateco, this means checking against ASTM B633 and any applicable OEM standards from customers like John Deere (JDM), Case (CNH), Caterpillar (CAT), and Parker Hannifin. Full documentation and traceability are maintained for quality audits. Defect rates below 0.2% and on-time delivery rates above 95% reflect what rigorous process control at every step makes possible.
Key Benefits of Zinc Barrel Plating for Small Parts
Zinc barrel plating’s position as the industry-standard corrosion protection method for small steel components isn’t accidental — it reflects a combination of advantages that few competing processes can match simultaneously.
Sacrificial galvanic protection Zinc corrodes before steel. Even scratched or damaged coatings continue protecting base metal through cathodic protection — a feature no paint or sealant can replicate.
Outstanding cost efficiency Processing thousands of parts simultaneously slashes labor costs per unit. Barrel plating is consistently the most economical finishing option for small, high-volume components.
Uniform coverage on complex geometry Tumbling motion exposes every surface — threads, blind holes, undercuts, recesses — to the plating solution from multiple angles, solving a fundamental challenge of small part finishing.
Flexible passivate options Clear, yellow, black, and olive drab chromate finishes let manufacturers meet specific appearance requirements or corrosion performance targets for different applications.
Meets major OEM specifications Quality barrel plating to ASTM B633, John Deere, Caterpillar, Case, and Parker Hannifin specs — with full documentation and traceability for audits.
RoHS and REACH compliant Modern trivalent chromate systems are free of hexavalent chromium, meeting environmental regulations while delivering superior corrosion performance.
Industries That Rely on Zinc Barrel Plating
Zinc barrel plating is a workhorse process used across virtually every sector of manufacturing that involves small steel components. Its combination of corrosion protection and production efficiency makes it the default choice for countless applications across multiple industries.
Automotive manufacturing Brake caliper clips, engine mount bolts, wheel nuts, and stampings — parts exposed to road salt, moisture, and temperature extremes. A single vehicle may contain hundreds of barrel-plated components.
Agricultural equipment Farm hardware faces direct soil contact, fertilizer chemicals, and freeze-thaw cycles. Zinc barrel plating provides the durability needed for years of field use without premature corrosion failure.
Construction hardware Anchors, bolts, connectors, and tie plates permanently exposed to outdoor weather need cost-effective corrosion protection that meets structural corrosion resistance requirements.
Industrial machinery Machine screws, internal brackets, and mechanical fasteners inside industrial equipment need protection against humidity, lubricants, and the vibration stresses of continuous operation.
Barrel Plating vs. Rack Plating: Which Is Right for Your Parts?
One of the most common questions manufacturers face when specifying zinc plating is whether parts should be barrel plated or rack plated. The answer depends on part size, production volume, cosmetic requirements, and dimensional tolerances. Here is a clear comparison.
| Factor | Barrel plating | Rack plating |
|---|---|---|
| Best part size | Small to medium components | Large or delicate parts |
| Production volume | High volume — thousands of parts | Lower volume, individual handling |
| Cost per part | ✔ Lower — most cost-efficient | ✗ Higher — more labor intensive |
| Cosmetic finish | Functional; minor contact marks possible | ✔ Superior for decorative parts |
| Complex geometry | ✔ Tumbling aids penetration | ✔ Controlled positioning |
| Thickness tolerance | Wider statistical range | ✔ Tighter — ±0.0001″ possible |
| Turnaround time | ✔ Faster for large batches | Varies by part complexity |
| Ideal applications | Fasteners, screws, stampings, clips | Large castings, decorative hardware |
Rule of thumbIf your parts are small, you’re producing them in high volumes, and cosmetic perfection isn’t the primary requirement — barrel plating is almost always the better choice. It delivers equivalent corrosion protection at a significantly lower cost per part.
Corrosion Resistance Performance and Industry Standards
The corrosion resistance delivered by zinc barrel plating isn’t a fixed quantity — it’s an engineered outcome that can be tuned precisely to the environment where the parts will operate. Three variables drive this performance: zinc coating thickness, the passivate system applied after plating, and any additional post-treatment sealers.
Zinc Coating Thickness
Thicker zinc deposits extend service life by delaying the onset of base steel exposure. Parts for indoor use in dry environments may require only minimal zinc thickness. Components exposed to road salts, coastal humidity, or agricultural chemicals need significantly heavier deposits. The primary industry standard governing this — ASTM B633 — defines four service condition numbers (SC 1 through SC 4) with corresponding minimum zinc thickness requirements for each exposure environment.
Passivate Selection
The chromate conversion coating applied over the zinc is the second major lever for controlling corrosion performance. Trivalent yellow passivate provides greater protection than clear passivate. Black and olive drab passivates serve specific applications where appearance and corrosion resistance must be balanced. A wax or sealer can be added as a third layer for maximum performance in the most demanding environments.
Salt Spray Testing
Salt spray testing — exposure to a saline mist inside a controlled chamber — is the standard laboratory method for evaluating and verifying corrosion resistance. Results are measured in hours to white corrosion (zinc degrading) and hours to red corrosion (base steel rusting). Well-executed zinc barrel plating with appropriate passivate treatment can achieve hundreds of hours of salt spray protection — and at Plateco, salt spray testing is integrated into quality management as both an internal QC tool and a customer service capability.
“We treat zinc plating as an extremely complex process demanding state-of-the-art technology, painstaking planning, obsessive quality control, and a tremendous amount of talent. Because our customers don’t come to us for excuses — they come to us for perfection.”
— Jim Schweich, Chief Executive Perfectionist, Plateco, Inc.
Frequently Asked Questions
Choosing the Right Zinc Barrel Plating Partner
Not all zinc plating operations are equal. When production schedules, part quality, and customer relationships depend on a plating partner, the wrong choice means defect rates, missed deliveries, and costly rework. Here is what separates a true specialist from a generalist finisher.
Specialization over generalism
A company that focuses exclusively on zinc plating will always outperform a general metal finisher that offers zinc as one of many services. Specialization means deeper process knowledge, more refined quality systems, and continuous investment in the one thing they do. Plateco, Inc. is a zinc-only specialist — it’s not a side service, it’s the entire business, since 1974.
Automation and process control
High-quality barrel plating depends on tight operational control — electrolyte chemistry balance, current density regulation, temperature management, and contamination prevention. Fully automated lines reduce human error and increase repeatability across every barrel run — essential for OEM production where consistency is non-negotiable. Plateco operates two fully automated zinc barrel electroplating production lines, each capable of handling up to 800 pounds per barrel.
Certifications and quality standards
ISO 9001 certification indicates a documented quality management system with defined processes for defect prevention, corrective action, and continuous improvement. Plateco holds ISO 9001:2015 certification — a foundation for the audit-ready quality control that OEM customers require. Every batch runs on a detailed work order — think of it as a precision recipe — that specifies every processing parameter so operators produce consistent results every single time.
Capacity and on-time delivery
A plating partner’s reliability is just as important as their coating quality. Plateco’s commitment is direct: on spec, on time — or it’s on us. A 95% on-time delivery rate and a defect rate below 0.2% back that commitment with measurable accountability.