If your manufacturing operation produces high volumes of small steel components — fasteners, clips, brackets, stampings, or machine screws — there is a good chance that zinc barrel electroplating is either already part of your supply chain or should be. It is one of the most widely used and cost-effective metal finishing processes in industrial manufacturing, providing reliable corrosion protection for enormous quantities of parts in a single production run.
But what exactly happens inside a barrel plating line? What are the steps involved, and why does each one matter? This guide walks you through the complete zinc barrel electroplating process from start to finish — covering the science behind the process, every critical step, the variables that affect quality, and how to know when barrel plating is the right choice for your parts.
Whether you are a procurement professional, a production engineer, or simply someone trying to understand what your plating supplier actually does, this complete overview will give you the foundational knowledge you need.
What Is Zinc Barrel Electroplating?
Zinc barrel electroplating is an electrochemical finishing process in which a protective zinc coating is deposited onto small steel parts in bulk quantities. The process uses electrical current passed through a zinc electrolyte solution to cause zinc ions to migrate and bond to the surface of the metal parts being processed.
What makes barrel plating unique compared to other electroplating methods is the delivery system: instead of mounting parts individually on racks or fixtures, barrel plating loads hundreds or thousands of small components into a perforated rotating barrel. That barrel is submerged in the plating bath, and as it slowly rotates, the tumbling action ensures that all parts are continuously exposed to the electrolyte solution and electrical current.
The result is a uniform zinc coating applied to every exposed surface of every part in the batch — simultaneously, efficiently, and at a cost per part that makes high-volume production economically practical.
Why Zinc? Understanding the Chemistry of Corrosion Protection
Before getting into the step-by-step process, it is worth understanding why zinc is used as the plating metal in the first place.
Steel corrodes through an electrochemical process called oxidation. When steel is exposed to oxygen and moisture, iron atoms give up electrons and form iron oxide — rust. Left unprotected, steel parts in outdoor, industrial, or high-humidity environments will degrade rapidly, losing structural integrity and dimensional accuracy.
Zinc protects steel through two complementary mechanisms:
Barrier Protection The zinc coating physically separates the steel from the corrosive environment. As long as the coating is intact, moisture and oxygen cannot reach the steel beneath.
Sacrificial (Galvanic) Protection This is zinc’s most important advantage. Zinc is less noble than steel on the electrochemical scale, meaning that when both metals are present in a corrosive environment, zinc corrodes preferentially. Even if the zinc coating is scratched, chipped, or partially damaged, the exposed steel will not corrode as long as zinc remains nearby. The zinc sacrifices itself to protect the steel — which is why zinc-coated parts continue to resist corrosion even after surface damage.
This sacrificial protection is what distinguishes zinc from purely decorative coatings and makes it the preferred choice for fasteners, automotive hardware, agricultural components, and construction connectors that face real-world exposure.
When Is Barrel Plating the Right Choice?
Zinc barrel electroplating is specifically designed for certain types of parts and production scenarios. Understanding where it excels — and where it is not the best fit — helps manufacturers make smarter finishing decisions.
Barrel Plating Is Ideal For:
- Small to medium-sized components (fasteners, screws, nuts, bolts, clips, stampings, springs, washers)
- High-volume production runs where cost efficiency is a priority
- Parts where cosmetic perfection is secondary to corrosion performance
- Components with complex geometries including threads, blind holes, and recesses
- Applications governed by standard corrosion resistance specifications such as ASTM B633
Barrel Plating Is Less Suited For:
- Large or heavy parts that cannot tumble safely without damage
- Highly cosmetic parts requiring a flawless appearance
- Delicate components that would be damaged by part-on-part contact during tumbling
- Parts requiring extremely tight dimensional tolerances, as the plating adds a small but measurable layer of zinc
For large, cosmetic, or delicate parts, rack electroplating — where each part is individually mounted and does not contact other parts during plating — is the preferred alternative. But for the vast majority of small industrial hardware, barrel plating delivers the right combination of performance, efficiency, and value.
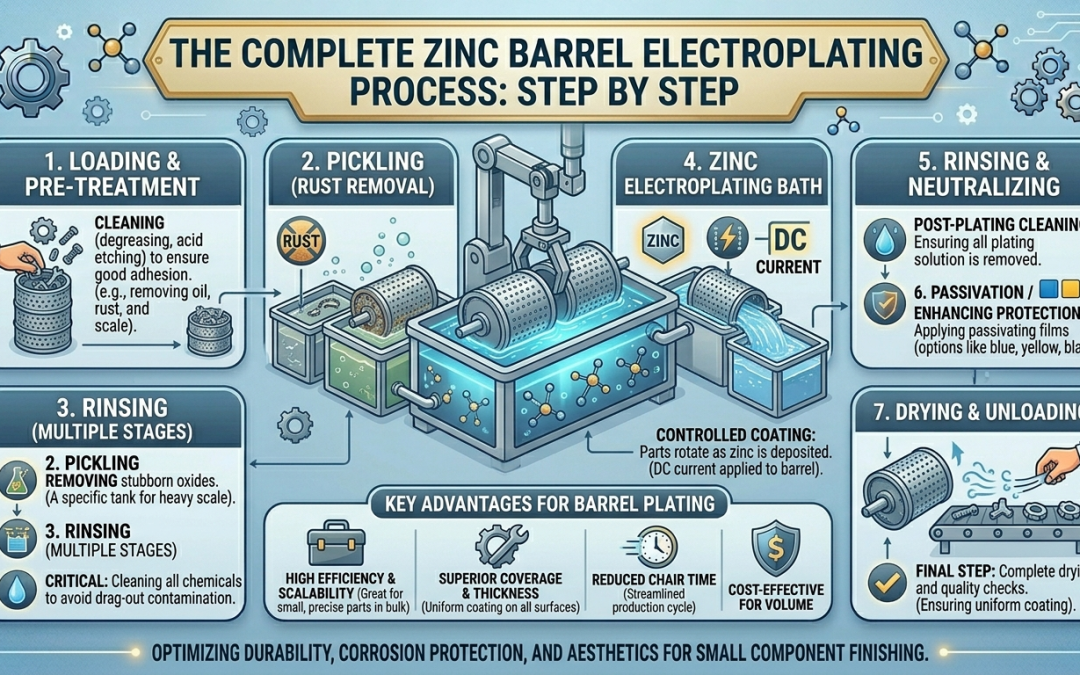
The Complete Zinc Barrel Electroplating Process: Step by Step
Step 1: Part Inspection and Pre-Processing Review
Before any parts enter the plating line, a thorough inspection and review process is essential. This step is often overlooked in descriptions of the plating process, but it is critically important.
The incoming parts are examined for:
- Base material verification (confirming the parts are the expected steel alloy)
- Surface condition assessment (looking for heavy scale, weld slag, existing coatings, or excessive rust that may require additional pre-treatment)
- Dimensional checks to establish baseline measurements before plating adds thickness
- Part geometry review to determine barrel loading density and rotation settings
This review sets the parameters for the entire plating process. A batch of heavily scaled stamped steel parts requires a different pre-treatment approach than a batch of precision-machined fasteners. Getting this step right prevents costly defects downstream.
Step 2: Bulk Loading into the Barrel
Once the pre-processing review is complete, parts are loaded in bulk into the plating barrel. The barrel itself is a perforated cylindrical or hexagonal container made from polypropylene or similar chemically resistant plastic. The perforations allow plating solution to flow freely through the barrel while keeping parts contained.
Barrel loading density matters significantly. Overloading the barrel can prevent adequate tumbling, resulting in parts that rest in fixed positions for extended periods and develop uneven plating — a condition called “nesting.” Underloading wastes capacity and increases cost per part. Experienced plating engineers determine the correct loading weight based on part size, geometry, and the specific plating parameters required.
At Plateco, Inc., barrel plating systems are engineered to process up to 800 pounds per barrel, with controlled rotation speeds that can be adjusted for delicate components that require gentler handling during tumbling.
Step 3: Alkaline Cleaning (Degreasing)
With parts loaded and the barrel sealed, the first active step in the plating process is alkaline cleaning, commonly called degreasing. This step removes oils, cutting fluids, forming lubricants, rust-preventive compounds, and other organic contamination from the part surfaces.
The barrel is submerged in a heated alkaline cleaning solution — typically a sodium hydroxide or sodium carbonate-based chemistry — and the parts tumble through the solution. Electrolytic cleaning, where electrical current is passed through the cleaning bath, is often used to enhance cleaning efficiency. The current causes the release of hydrogen or oxygen gas at the part surfaces, which provides a mechanical scrubbing action that dislodges stubborn contamination.
Why is this step so critical? Zinc cannot bond properly to a contaminated surface. If oils or organic compounds remain on the steel, the zinc will deposit over them rather than directly onto the metal — resulting in poor adhesion, blistering, or flaking of the coating after plating. The quality of the entire plating job depends on how thoroughly the cleaning steps are performed.
Step 4: Rinsing After Alkaline Cleaning
After degreasing, the barrel moves through one or more rinse stations. Rinsing removes the alkaline cleaning chemistry from the part surfaces and the barrel before the parts advance to the next step.
Rinsing seems simple, but it is operationally important for two reasons. First, alkaline carry-over into the acid pickling bath (the next step) would neutralize the acid and reduce its effectiveness. Second, inadequate rinsing can leave chemical residues on part surfaces that interfere with subsequent processing steps.
Modern automated plating lines use counter-current rinsing systems, where parts move through multiple rinse tanks in sequence, with fresh water entering the system at the final rinse and flowing backward through earlier tanks. This conserves water while maintaining rinse quality — an important consideration for both environmental compliance and operational cost.
Step 5: Acid Pickling (Oxide Removal)
Even after thorough alkaline cleaning, steel parts still carry a layer of iron oxide on their surfaces. This oxide layer forms naturally as steel is exposed to air and moisture during manufacturing, storage, and shipping. If not removed, it creates a barrier between the steel and the zinc coating, preventing proper adhesion.
Acid pickling uses a dilute hydrochloric acid or sulfuric acid solution to dissolve the iron oxide layer and expose the clean, reactive steel surface beneath. The barrel tumbles through the acid solution for a controlled period — long enough to remove oxides without over-etching the base metal.
Over-pickling is a real risk. If parts remain in the acid too long, the steel itself begins to dissolve, causing hydrogen embrittlement — a condition where atomic hydrogen diffuses into the steel crystal structure and can cause premature fracture under load. For high-strength steel parts, particularly those used in critical fastening applications, careful control of pickling time and chemistry is essential, and post-plating baking (hydrogen embrittlement relief) may be required.
Step 6: Rinsing After Acid Pickling
Another rinse stage follows acid pickling to remove acid residue before the parts enter the plating bath. Acid carry-over into the plating electrolyte would alter its chemistry and compromise the quality of the zinc deposit.
The rinse water from this stage must be treated before discharge as part of environmental compliance protocols, since it contains dissolved metals and acid residues.
Step 7: Zinc Electrodeposition (The Plating Step)
This is the core of the entire process — the step where zinc is actually deposited onto the steel parts. The parts, now thoroughly cleaned and free of surface oxides, enter the zinc plating bath inside the rotating barrel.
The plating bath contains a zinc electrolyte solution — most commonly an alkaline zinc cyanide or, more commonly today, an alkaline non-cyanide zinc system or an acid zinc chloride system. Each chemistry has different characteristics in terms of plating speed, bath stability, coating distribution, and environmental impact.
Inside the bath, the electrical circuit is established:
- The steel parts act as the cathode (negative electrode)
- Zinc anodes (either bars or balls in anode baskets) act as the anode (positive electrode)
- Direct electrical current flows through the electrolyte solution between the anodes and the tumbling parts
At the cathode (the parts), zinc ions in solution gain electrons and are reduced to metallic zinc, depositing as a solid layer on the steel surface. At the anode, metallic zinc dissolves into the solution to replenish the zinc ions consumed at the cathode.
The tumbling action of the barrel continuously shifts parts relative to each other and relative to the electrical field, ensuring that no single area remains stationary long enough to receive more or less zinc than surrounding areas. This is what produces the uniform coating distribution that barrel plating is known for — including coverage on threads, inside blind holes, around recessed features, and on all sides of complex geometries.
Key process variables controlled during this step include:
- Current density — the amount of electrical current per unit area of part surface. Too low produces a thin, uneven coating; too high causes rough, powdery, or burned deposits.
- Bath temperature — affects zinc ion mobility and deposition rate
- Electrolyte chemistry — zinc concentration, brighteners, and leveling agents must be maintained within strict limits
- Barrel rotation speed — affects coating uniformity and part damage potential
- Plating time — determines final coating thickness
At Plateco, Inc., automated chemistry monitoring systems continuously track and adjust the electrolyte composition, ensuring consistent coating quality from barrel to barrel and shift to shift. This level of process control is what separates a precision plating operation from a commodity shop.
Step 8: Rinsing After Plating
When the required plating time is complete, the barrel is removed from the zinc bath and rinsed to remove electrolyte solution from the part surfaces and the barrel itself. This step prevents chemical carry-over into the post-plating treatment stages.
Step 9: Chromate Conversion Coating
Fresh zinc plating, while effective at corrosion protection, is relatively soft and develops a dull, white corrosion product (zinc oxide) fairly quickly when exposed to moisture. To enhance both the corrosion resistance and the appearance of the zinc coating, parts typically receive a chromate conversion coating immediately after plating.
Chromate conversion coating is an electrochemical treatment that reacts with the zinc surface to form a thin, complex chromium compound film. This film provides several important benefits:
- Enhanced corrosion resistance — chromate-treated zinc parts significantly outperform untreated zinc in salt spray testing
- Sealing of micro-pores in the zinc deposit
- Color and appearance — different chromate chemistries produce different colors, allowing visual identification of corrosion protection level and specification compliance
The most common chromate finish colors available include:
Clear (Blue-Bright) The thinnest chromate treatment, producing a bright, clear to slightly blue finish. Provides moderate corrosion resistance improvement over bare zinc.
Yellow (Iridescent) A thicker chromate film with a distinctive gold-yellow iridescent appearance. Provides significantly higher corrosion resistance than clear chromate.
Black A specialty chromate chemistry producing a uniform black finish. Used for aesthetic requirements or applications where light reflection must be minimized.
Olive Drab Used primarily in military and defense applications where specific corrosion performance and appearance standards must be met.
Custom Finishes Specialty coatings and post-treatment combinations can be engineered to meet specific OEM or industry standards.
It is important to note that modern environmental regulations have driven the transition from hexavalent chromium (Cr6+) chromates to trivalent chromium (Cr3+) chemistries in many applications. Trivalent chromates are less hazardous while providing comparable or superior corrosion performance.
Step 10: Rinsing After Chromate Treatment
A final rinse removes excess chromate chemistry from the parts before the sealing or drying step.
Step 11: Sealing (Optional but Recommended)
After chromate treatment, parts may receive a topcoat sealer. Sealers are thin organic or inorganic coatings applied over the chromate film that fill remaining micro-porosity and further extend corrosion resistance.
Sealers are particularly valuable in applications where maximum salt spray performance is required, where parts will be exposed to aggressive environments, or where customer specifications demand performance above what chromate alone can provide.
Step 12: Drying and Unloading
The final step in the plating line is drying — removing water from the part surfaces to prevent water staining and to set the chromate and sealer films. Parts are typically dried using heated forced-air dryers or centrifugal spin-drying systems.
After drying, parts are unloaded from the barrel, inspected for coating quality and appearance, and prepared for packaging and shipment.
Quality Control Throughout the Process
A zinc barrel electroplating operation is only as good as its quality control systems. At a precision plating facility like Plateco, Inc., quality monitoring is not a final inspection — it is integrated throughout every stage of the process.
Key quality control activities include:
Coating Thickness Measurement Zinc coating thickness is measured using magnetic induction gauges on finished parts. Thickness requirements are defined by specifications such as ASTM B633, which establishes minimum thickness classes based on service environment severity.
Salt Spray Testing Parts are subjected to standardized salt spray exposure (per ASTM B117) to verify that the coating system meets corrosion resistance requirements before shipment.
Visual Inspection Trained inspectors examine finished parts for plating defects including rough deposits, bare spots, burning, blistering, or poor chromate uniformity.
Chemistry Analysis The plating bath, cleaning solutions, and chromate chemistry are regularly sampled and analyzed — either in-house or by laboratory services — to verify that all chemistry is within specification.
Documentation and Traceability Production records document processing parameters, chemistry results, thickness measurements, and salt spray data for each batch, supporting quality audits and customer compliance requirements.
Plateco, Inc. operates under ISO 9001:2015 certification, meaning that every aspect of the quality management system — from incoming part inspection through final shipment — is governed by documented procedures and subject to regular internal and external audits.
Zinc Barrel Plating and Environmental Compliance
Modern zinc barrel electroplating operations must meet strict environmental regulations governing the handling, treatment, and disposal of process chemicals and wastewater.
Key environmental considerations include:
Wastewater Treatment Rinse waters and spent chemistry from the plating line contain dissolved zinc, chromium, and other metals that must be treated before discharge. Treatment systems typically involve pH adjustment, precipitation of metals as hydroxides, and filtration before discharge or recycling.
Hexavalent Chromium Restrictions Regulations in many jurisdictions restrict or prohibit the use of hexavalent chromium in chromate coatings. Trivalent chromate systems have largely replaced hex-chrome in compliant operations.
Sludge Disposal Metal hydroxide sludge from wastewater treatment must be disposed of as a regulated waste through licensed hazardous waste contractors.
A responsible zinc plating partner maintains all required environmental permits, operates compliant wastewater treatment systems, and provides documentation of environmental compliance on request.
Industries That Rely on Zinc Barrel Electroplating
Zinc barrel electroplating serves a broad range of industries wherever small steel components require reliable corrosion protection:
Automotive Manufacturing Fasteners, clips, brackets, mounting hardware, and stampings exposed to road salt, moisture, and temperature cycling. Automotive specifications often require specific salt spray performance verified by standardized testing.
Agricultural Equipment Outdoor hardware including bolts, nuts, pins, and connectors subjected to soil contact, moisture, fertilizer exposure, and UV radiation.
Construction and Infrastructure Anchors, concrete fasteners, structural connectors, and hardware exposed to weather, concrete alkalinity, and long service life requirements.
Industrial Machinery Internal fasteners, machine screws, and mechanical components where reliable corrosion resistance extends service intervals and reduces maintenance.
Consumer Products and Appliances Hardware components in products where corrosion resistance, appearance, and cost efficiency must all be balanced.
Barrel Electroplating vs. Rack Electroplating: Choosing the Right Process
Both barrel and rack electroplating produce high-quality zinc coatings, but they are designed for different types of work.
Barrel electroplating excels when parts are small, quantities are high, and cost efficiency is the priority. The ability to process thousands of parts simultaneously in a single barrel makes it dramatically more economical on a per-part basis than rack plating for small components.
Rack electroplating is preferred when parts are large, heavy, or fragile, when cosmetic appearance is critical, or when extremely tight dimensional tolerances must be maintained and part-on-part contact cannot be permitted. Each part is individually mounted on a rack, ensuring no contact damage and allowing more precise control of coating distribution on critical surfaces.
For most fasteners and small hardware, barrel plating is the obvious choice. When in doubt, your plating partner should be able to evaluate your parts and recommend the most appropriate process.
Frequently Asked Questions About Zinc Barrel Electroplating
How thick is the zinc coating on barrel-plated parts?
Coating thickness depends on the specification and service environment. ASTM B633 defines several service condition classes ranging from SC1 (mild) at 5 micrometers minimum to SC4 (very severe) at 25 micrometers minimum. Your plating supplier should confirm the thickness class required for your application.
How long will zinc barrel-plated parts resist corrosion?
This depends on coating thickness, chromate treatment type, and service environment. In standard salt spray testing, properly processed zinc-plated parts with yellow chromate can achieve hundreds of hours of corrosion resistance. Real-world service life varies based on actual environmental conditions.
Can high-strength steel parts be barrel plated?
Yes, but with precautions. High-strength steel is susceptible to hydrogen embrittlement from the acid pickling step. Parts above a certain hardness threshold (typically 40 HRC per ASTM F1941) require baking after plating to relieve hydrogen before the coating is stressed.
What is the difference between alkaline and acid zinc plating?
Alkaline zinc systems generally provide better coating distribution across complex geometries and are preferred for threaded parts. Acid zinc systems plate faster and produce brighter deposits but may have less uniform distribution on complex shapes. Your plating engineer can recommend the appropriate system for your parts.
Does barrel plating affect the dimensions of my parts?
Yes, to a small degree. Zinc plating adds material to the part surface. For standard coating thicknesses, this addition is typically a few micrometers per surface — negligible for most applications but important for parts with very tight dimensional tolerances. Discuss tolerances with your plating supplier before processing.
Conclusion: Why the Process Details Matter
Zinc barrel electroplating looks simple from the outside — load parts, plate them, ship them out. But as this step-by-step overview makes clear, the process involves a carefully orchestrated sequence of chemical and electrochemical steps, each of which must be executed correctly for the final coating to perform as required.
Every stage — from alkaline cleaning through acid pickling, zinc deposition, chromate treatment, and final quality inspection — affects the adhesion, thickness, uniformity, and corrosion resistance of the finished coating. Skipping steps, using out-of-specification chemistry, or operating without adequate process controls produces defective parts that fail in service.
That is why choosing a zinc plating partner with deep process knowledge, disciplined quality systems, and a genuine commitment to getting it right every time is so important. At Plateco, Inc., zinc barrel electroplating is not a side service — it is our entire focus. Our automated barrel lines, ISO 9001:2015 quality systems, and unwavering commitment to delivering parts on spec and on time make us the trusted partner for manufacturers across Wisconsin and beyond.
On Spec. On Time. Or It’s On Us.
Ready to discuss your zinc barrel electroplating requirements? Contact Plateco, Inc. at (608) 524-8241 or request a quote online to speak with our team about your parts, volumes, and corrosion resistance requirements.