Walk through any manufacturing facility and you will find steel parts everywhere — brackets, fasteners, shafts, housings, connectors, and structural hardware of every shape and size. What you will not always see is the thin but enormously consequential coating on many of those parts that determines whether they last three years or thirty.
Zinc plating is that coating for the majority of industrial steel components. It is not glamorous. It is not complicated to understand at a conceptual level. But it is one of the most important finishing decisions any manufacturer makes, and choosing the wrong process — or the wrong plating partner — costs real money in failed parts, premature replacements, warranty claims, and production downtime.
This guide gives you a practical, honest foundation in zinc plating for industrial machinery: what it does, why it works, how the different processes compare, what variables actually matter, and how to evaluate whether your current approach is serving your operation as well as it should.
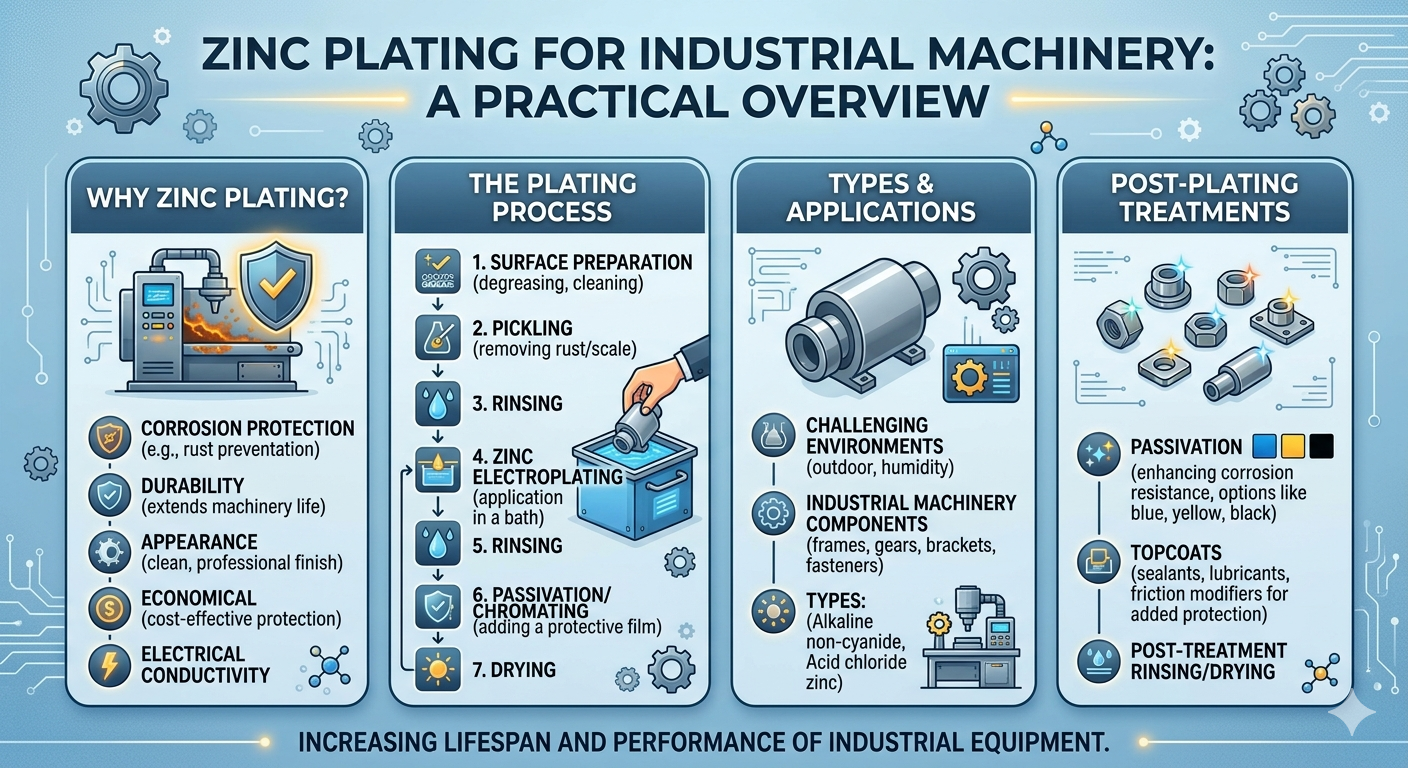
What Zinc Plating Actually Does — and Why It Works
Steel corrodes. That is not a design flaw or a material weakness that modern engineering has somehow failed to solve — it is a fundamental electrochemical reality. Iron, the primary constituent of steel, readily oxidizes in the presence of oxygen and moisture, producing the familiar reddish-brown iron oxide we call rust. In industrial environments, where machinery components encounter humidity, temperature cycling, chemical exposure, road salt, agricultural materials, or simple outdoor weathering, this oxidation process accelerates dramatically.
Zinc plating interrupts that process through two distinct mechanisms, which is what makes it so effective compared to purely barrier coatings like paint or lacquer.
Barrier Protection
The first mechanism is physical. The zinc layer deposited on the steel surface creates a barrier between the steel and the corrosive environment. Oxygen and moisture cannot reach the iron substrate as long as the zinc coating remains intact. This is straightforward barrier protection — the same principle that underlies any coating — and it is effective as long as the coating has not been scratched, chipped, or worn through.
Sacrificial (Cathodic) Protection — The Real Advantage
The second mechanism is where zinc plating genuinely separates itself from most other coating approaches. Zinc is electrochemically more active than iron, which means that when zinc and iron are in contact in the presence of an electrolyte (moisture), zinc preferentially oxidizes — it corrodes in place of the iron.
This sacrificial protection means that even if the zinc coating is scratched and bare steel is exposed, the surrounding zinc will continue to corrode sacrificially, protecting the steel at the scratch until the local zinc supply is exhausted. A painted part with a scratch corrodes at the scratch and progresses. A zinc-plated part with a scratch is still protected — the zinc sacrifices itself so the steel does not have to.
This is why Plateco Inc., which focuses exclusively on zinc plating, describes the corrosion progression in precise terms: the outer passivate and sealer protect the zinc from white corrosion, while the zinc itself corrodes sacrificially to protect the underlying steel from red corrosion. White corrosion — the powdery zinc oxide that appears on a zinc-plated surface — is not a sign of coating failure. It is a sign the zinc is doing exactly what it is designed to do.
The Three-Layer System
A fully specified zinc plating system consists of three engineered layers, each with a specific role:
Layer 1 — Zinc. The primary sacrificial layer deposited directly on the steel. Its thickness determines the duration of protection before the steel is exposed.
Layer 2 — Trivalent passivate (chromate conversion coating). A chemical conversion coating applied over the zinc that slows the rate of zinc corrosion, extending service life substantially. Passivates also determine the visual appearance of the finished part — clear/blue, yellow, black, or olive drab finishes all result from specific passivate chemistries.
Layer 3 — Wax or sealer. An optional but frequently specified topcoat that provides an additional barrier against moisture and atmospheric attack on the passivate layer, further extending corrosion performance.
Understanding this three-layer system is essential for interpreting corrosion resistance specifications, which are almost always expressed in hours of salt spray resistance to red rust. A bare zinc coating without passivate or sealer will fail to red rust far sooner than a fully sealed system of equivalent zinc thickness.
Why Industrial Machinery Components Need Zinc Plating
Industrial machinery operates in environments that accelerate corrosion at rates that would seem extreme in everyday contexts. Consider what a typical agricultural equipment component experiences over a single season: soil moisture, fertilizer chemicals, rain, temperature swings from below freezing to summer heat, UV exposure, abrasion from soil and debris, and periodic washing with high-pressure water. A structural fastener in that environment without adequate corrosion protection might last a single season before red rust compromises its structural integrity.
The industrial categories where zinc plating provides measurable protection against real operational losses include:
Agricultural equipment. Linkage components, structural hardware, and fasteners on tractors, combines, planters, and tillage equipment are subjected to some of the harshest corrosive environments in any industry. Soil chemistry, moisture retention, chemical fertilizers, and prolonged outdoor storage combine to accelerate corrosion aggressively.
Automotive and heavy vehicle components. Road salt — used extensively in cold-weather regions like Wisconsin — is one of the most corrosive substances that industrial components routinely encounter. Brackets, clips, fasteners, mounting hardware, and structural components on vehicles must withstand constant exposure to salt spray and wet-dry cycling.
Construction hardware. Anchors, bolts, connectors, and structural brackets used in construction projects are often buried in concrete, embedded in soil, or permanently exposed to outdoor weathering. Corrosion failure in these applications is not just a cost problem — it is a structural integrity problem.
Industrial machinery and equipment. Machine screws, internal hardware, brackets, and assembly components in manufacturing equipment must maintain dimensional and structural integrity over long service lives in environments that may include industrial humidity, chemical exposure, and repeated washdown.
General OEM manufacturing. Any original equipment manufacturer producing steel components for assembly into larger systems — whether that is HVAC equipment, material handling systems, power transmission assemblies, or any other industrial product — relies on zinc plating to deliver the corrosion life their customers expect.
The Three Zinc Plating Processes: What Differentiates Them
Not all zinc plating is the same process, and not every process is the right choice for every part. There are three primary methods used in industrial zinc plating, each with specific characteristics, advantages, and ideal applications.
Zinc Barrel Electroplating
Barrel electroplating is the high-volume workhorse of the zinc plating industry. Large quantities of small steel components — fasteners, stampings, clips, machine screws — are loaded into a perforated rotating barrel that is immersed in a zinc electrolyte solution. As electrical current flows through the system, zinc ions deposit onto the tumbling parts.
The barrel’s continuous rotation serves two functions: it ensures that all surfaces of each part are exposed to the electrolyte (and therefore receive zinc deposition), and it eliminates the need to individually mount or handle each part, which makes the process extremely cost-efficient at high volumes.
Barrel plating is the right choice when:
- Parts are small (typically fitting within a defined volume that allows effective tumbling)
- Production volumes are high — hundreds or thousands of identical parts per run
- Cosmetic perfection is secondary to functional corrosion protection
- Cost efficiency is a priority
- Parts do not have features that would be damaged by tumbling contact between pieces
Plateco’s barrel electroplating systems are capable of processing up to 800 pounds per barrel load, with fully automated production lines that maintain consistent chemistry monitoring and deposition control. Seven cleaning stages — including heated soaps, acids, and proprietary cleaning processes — ensure that even heavily contaminated parts from processes like heat treating or welding are properly prepared before zinc deposition begins. Proper cleaning is not a preliminary step — it is the foundation on which zinc adhesion depends.
Zinc Rack Electroplating
Rack electroplating addresses a fundamental limitation of barrel plating: parts that are too large, too complex, too fragile, or too cosmetically sensitive to survive tumbling in a barrel need individual handling.
In rack plating, each component is individually mounted on a specialized rack fixture that maintains its position and electrical contact throughout the plating process. The rack is then immersed in the plating bath, and electrical current causes zinc deposition on each individually supported part.
The advantages of rack plating over barrel plating are concentrated in three areas: coating thickness control, surface finish quality, and the ability to handle large or complex geometry parts.
Thickness control. Because each part is individually mounted and the current density at each part’s surface can be precisely controlled, rack plating allows engineers to specify and achieve exact coating thicknesses. This is critical for assemblies with tight dimensional tolerances, where coating thickness directly affects fit and function.
Surface finish quality. Without the surface-to-surface contact that occurs in barrel tumbling, rack-plated parts emerge with smoother, brighter finishes. This matters for visible components, exterior hardware, or parts whose appearance is a quality criterion.
Geometry handling. Large brackets, structural components, machined parts, and assemblies with features that require individual attention — rack mounting allows custom fixture design that accommodates unusual geometry and ensures consistent coverage.
Rack plating is the appropriate choice when:
- Parts are large or heavy
- Precise coating thickness is specified to engineering tolerances
- Cosmetic finish quality is a customer requirement
- Parts have features that would be damaged by tumbling contact
- Individual part traceability or inspection is required
Plateco’s rack plating systems include engineering planning that designs custom rack fixtures for each part type, ensuring consistent positioning, electrical contact, and current distribution across every production run. The company’s Engineering Planning Department considers over 80 sets of tooling options, surface area calculations, current density requirements, and geometry-specific quality risk factors for each new part before a work order is created.
Zinc Mechanical Galvanizing
Mechanical galvanizing occupies a distinct position in the zinc plating spectrum — one that is frequently misunderstood and underspecified. It is not an electroplating process at all. Instead of using electrical current to deposit zinc ions onto steel surfaces, mechanical galvanizing uses physical force: zinc powder is tumbled with parts in a rotating drum, and the mechanical energy of the tumbling process embeds zinc particles directly into the steel surface.
The result is a zinc coating that is typically thicker than electroplated zinc and bonded differently — through mechanical embedment rather than electrochemical deposition.
The practical consequences of this difference are significant:
No hydrogen embrittlement risk. Electroplating processes introduce hydrogen into the steel during the electrochemical reduction reaction. In high-strength steel components, this absorbed hydrogen can cause hydrogen embrittlement — a weakening of the metal that can lead to delayed cracking under load. Mechanical galvanizing introduces no hydrogen, making it the safe choice for high-strength fasteners, springs, and structural hardware where embrittlement is a genuine engineering concern.
Thicker coatings without heat distortion. Mechanical galvanizing can achieve coating thicknesses that would be impractical with standard electroplating, without exposing parts to the high temperatures of hot-dip galvanizing. This matters for components that have been heat-treated to specific strength values — hot-dip galvanizing temperatures can affect the metallurgical properties of some heat-treated steels. Mechanical galvanizing delivers heavy coatings without thermal risk.
Consistent coverage on complex features. The tumbling action of mechanical galvanizing penetrates threads, blind holes, recesses, and undercuts effectively, delivering consistent zinc coverage across complex geometries.
Mechanical galvanizing is the specified choice for:
- High-strength fasteners (Grade 8 bolts, high-tension structural hardware) where hydrogen embrittlement must be avoided
- Large structural hardware requiring heavy zinc coatings
- Anchor bolts, heavy connectors, and mining or construction components exposed to aggressive environments
- Applications where coating thickness requirements exceed what is practical with standard electroplating
What “Specification” Actually Means in Zinc Plating
When a drawing or purchase order specifies “zinc plating,” that is typically the beginning of the specification, not the end of it. A complete zinc plating specification defines several parameters that determine whether the finished coating actually performs as required.
Coating Thickness
Zinc coating thickness is typically expressed in microns (µm) or thousandths of an inch. The governing specification in the United States for most zinc electroplating applications is ASTM B633, which defines four service condition classes:
- SC1 (Mild): 5 µm minimum — indoor or controlled environments
- SC2 (Moderate): 8 µm minimum — general outdoor exposure
- SC3 (Severe): 12 µm minimum — harsh outdoor exposure
- SC4 (Very Severe): 25 µm minimum — very aggressive environments
Specifying the wrong service class — either by underestimating the severity of the service environment or by defaulting to the lowest class to reduce cost — is one of the most common sources of premature coating failure in industrial applications.
Plateco meets ASTM B633 as a baseline specification and also complies with more demanding specifications including ASTM B695 for mechanical galvanizing, CAT 1E0397, JDM F15/F22/F23, JS-500 (requiring 500 hours to red rust), and a range of OEM-specific standards including those from Toro and John Deere. These specifications represent the real-world performance targets that major equipment manufacturers have determined are necessary based on actual field experience with their products.
Passivate Chemistry and Finish Color
The chromate conversion coating applied over the zinc layer is not cosmetic — it is functional. Different passivate chemistries offer different levels of corrosion resistance. In general terms:
- Clear/blue passivate provides a baseline level of protection and a clean, professional appearance
- Yellow chromate provides enhanced corrosion resistance compared to clear passivate
- Black trivalent passivate offers a specific aesthetic finish with good corrosion performance
- Olive drab is specified for certain military and specialty industrial applications
The choice of passivate should be driven by the corrosion resistance requirement of the application, not by cost or default. A part specified for 200 hours of salt spray resistance to red rust needs a different passivate system than a part specified for 500+ hours.
Sealer Specification
Adding a wax or polymer sealer over the passivate layer extends corrosion performance substantially. For parts that need to meet aggressive salt spray specifications — particularly those at 500 hours or more to red rust — a sealer is typically required. The sealer also adds some lubricity to fastener surfaces, which can be operationally important for assembly applications where torque-tension relationships matter.
The Cleaning Problem That Ruins More Zinc Plating Than Anything Else
The most frequent cause of zinc plating failure is not inadequate coating thickness or the wrong passivate chemistry. It is inadequate surface preparation before plating begins.
Zinc electroplating adheres to steel because zinc bonds to the iron in the steel surface. Any contamination that covers that iron surface — oil, cutting fluid, heat treat scale, weld flux, rust, or any other material deposited during manufacturing — physically prevents zinc adhesion. The result is poor adhesion, blistering, bare spots, or coating that appears acceptable but fails under the first exposure to corrosive conditions.
Many zinc plating operations expect their customers to pre-clean parts before sending them for plating. This adds cost and logistics complexity to the customer’s operation and introduces variability — parts cleaned in different shops, to different standards, with different equipment, on different schedules are not going to arrive at consistent cleanliness levels.
Plateco has invested in cleaning capabilities that eliminate the pre-cleaning requirement. Seven integrated cleaning stages — including heated soaps, multiple acid stages, and proprietary cleaning steps developed specifically for the most stubborn industrial contaminants — are built directly into the fully automated production lines. Parts arrive from manufacturing. They go into the line. The cleaning happens as part of the plating process, not as a separate external step.
This integration is not just a convenience. It is a quality control mechanism. When cleaning and plating happen in the same controlled automated system, the variability introduced by external pre-cleaning disappears. Every part receives the same cleaning process in the same sequence under the same controlled conditions, every time.
Process Control: What Separates Acceptable From Excellent
Zinc plating is not technically complex to perform at a basic level. Running a barrel plating line and producing zinc-coated parts that pass a visual inspection is not difficult. Producing zinc-coated parts that consistently meet engineering specifications across thousands of parts per day, with defect rates below 0.15%, run after run, requires something entirely different.
Chemistry Monitoring and Control
A zinc plating bath is a chemical system that changes continuously during production. Zinc concentration, pH, brightener levels, temperature, contamination — all of these parameters drift during operation and must be actively monitored and adjusted. An electrolyte bath that is outside its optimal parameters does not plate consistently. Thickness varies. Brightness varies. Adhesion varies.
Plateco maintains a dedicated lab department — three specialists whose entire responsibility is monitoring and maintaining plating bath chemistry within defined parameters. Temperature, pH, and chemical concentrations are tracked and controlled throughout every production shift. The bath is not checked periodically and adjusted reactively. It is monitored continuously and maintained proactively.
Engineering Planning
When a new part enters Plateco’s system for the first time, it does not simply get dropped into an existing line setup. The Engineering Planning Department analyzes the part’s geometry, material, specification requirements, and quality-risk factors before creating a work order that specifies exactly how the part will be processed — which plating line, which tooling configuration, what surface area per rack or barrel load, what current density, what cleaning sequence.
This work order functions as a baking recipe: every variable is defined in advance, every production decision is codified, and operators execute a specified process rather than making judgment calls on the floor. The result is that the 500th run of a given part looks like the first run, which looks like the 50th run.
Continual Improvement
Even with comprehensive planning and chemistry control, defects occur. What matters is how an organization responds to them. At Plateco, a daily 9 AM continual improvement meeting brings all department heads together on the production floor to review every reject from the previous day. The meeting does not end until the root cause of each defect is understood and a corrective action is in place.
This is not a quality management philosophy statement — it is a daily operational practice, conducted in public view on the production floor. The reject rate for the second quarter of 2025 was 0.13%. That number is the result of that daily process applied relentlessly over many years.
How to Evaluate Whether Your Current Zinc Plating Is Performing
If you are currently using zinc-plated components in your machinery and are not confident that your plating is performing to specification, here are the practical questions to ask.
Are you receiving a complete specification or just “zinc plate”?
If your drawings specify “zinc plate” without a service condition class, passivate type, or corrosion performance requirement, you are not specifying the coating you need. You are specifying a generic process and hoping it is adequate.
Do your parts arrive with pre-cleaning requirements?
If your plater requires you to clean parts before shipping, you are managing a process variable that your plater should own. Platers with adequate cleaning capability eliminate that burden.
What is your plater’s documented defect rate?
If your plater cannot tell you their defect rate — or if they measure it differently than you expect — that is informative. A plater confident in their quality posts their reject rate publicly. Plateco’s second quarter 2025 reject rate of 0.13% is displayed on their homepage.
Are your parts failing in the field before expected service life?
Premature field failures are the most expensive signal that something is wrong with coating specification or process quality. Understanding whether the failure mode is insufficient zinc thickness, wrong passivate for the environment, adhesion failure, or something else determines what needs to change.
Can your plater meet your specification certifications?
If your customers or OEM partners require compliance with ASTM B633, ASTM B695, or industry-specific specifications like JDM standards or CAT specifications, your plater needs documented, auditable compliance with those standards — not just an assertion that they can hit the numbers.
A Practical Decision Framework: Choosing the Right Process
The table below summarizes the practical decision criteria for selecting among the three zinc plating processes for industrial machinery components.
| Criterion | Barrel Electroplating | Rack Electroplating | Mechanical Galvanizing |
| Part Size | Small to medium | Medium to large | Medium to large |
| Production Volume | High | Low to medium | Medium to high |
| Thickness Control | Good | Excellent | Very good (thicker coatings) |
| Surface Finish | Functional | Bright, cosmetic | Matte, dense |
| Complex Geometry | Good (tumbling helps threads) | Excellent (custom fixtures) | Very good (tumbling action) |
| Hydrogen Embrittlement Risk | Possible | Possible | None |
| Ideal Applications | Fasteners, clips, stampings | Brackets, housings, visible parts | High-strength hardware, anchor bolts |
| Relative Cost | Lowest per part | Higher per part | Moderate |
| Key Standard | ASTM B633 | ASTM B633 | ASTM B695 |
For most industrial machinery fasteners and small hardware, barrel electroplating is the default starting point. For larger structural components, complex machined parts, or parts with cosmetic requirements, rack electroplating is the appropriate specification. For high-strength hardware where hydrogen embrittlement must be excluded, or for components requiring heavy coatings in very aggressive service environments, mechanical galvanizing is the right engineering choice.
Frequently Asked Questions
What is the difference between zinc plating and galvanizing?
Zinc plating typically refers to electroplating — using electrical current to deposit zinc on steel surfaces — or mechanical galvanizing, which uses mechanical force to embed zinc powder. Hot-dip galvanizing is a separate process that dips steel into molten zinc at high temperatures, producing a much thicker coating suited to structural steel and heavy construction applications. The high temperatures of hot-dip galvanizing can affect heat-treated components, which is one reason mechanical galvanizing is often preferred for high-strength hardware.
How long does zinc plating last?
Service life depends on coating thickness, passivate system, sealer presence, and the severity of the service environment. A properly specified zinc plating system for moderate outdoor exposure might provide five to ten years of protection before red rust appears. In highly aggressive environments — road salt, continuous moisture, chemical exposure — service life is shorter, and thicker coatings or more aggressive passivate systems are required.
Does zinc plating affect part dimensions?
Yes, but typically in a predictable and manageable way. Standard zinc electroplating adds 5 to 25 microns per surface, depending on specification. For most industrial hardware, this is negligible. For precision machined parts with tight assembly tolerances, coating thickness must be factored into the dimensional specification. This is one of the reasons precise thickness control — achievable with rack plating — matters for tolerance-critical components.
Can zinc-plated parts be welded after plating?
Welding through zinc-plated surfaces produces zinc oxide fumes, which require adequate ventilation and may require respiratory protection. From a quality standpoint, welding damages the zinc coating locally and requires touch-up or reprocessing of the affected areas to maintain corrosion performance. The preferred practice is to plate after welding, not before.
What causes white rust on zinc-plated parts?
White corrosion — the powdery white deposit on zinc surfaces — is zinc oxide, produced when zinc oxidizes. It is the intended behavior of the sacrificial zinc layer and is not a failure of the coating. It becomes visible when the passivate layer has been consumed and the zinc itself begins to corrode. White rust indicates the zinc is doing its job. Red rust indicates that the zinc has been fully consumed and the underlying steel is now corroding — that is when component integrity is at risk.
Conclusion
Zinc plating for industrial machinery is not a commodity finishing step — it is an engineered corrosion protection system whose performance depends on process selection, specification accuracy, cleaning quality, chemistry control, and plating partner expertise.
The difference between a zinc plating operation that delivers 0.13% defect rates and 95% on-time delivery and one that delivers chronic rejects and missed schedules is not luck. It is engineering discipline, process investment, and an organizational commitment to treating zinc plating as the complex, consequential process it actually is.
For industrial manufacturers in Wisconsin and across the country, understanding these fundamentals puts you in a position to specify better, evaluate your current process more critically, and make better decisions about the coatings protecting your components.
Plateco Inc. — located in Reedsburg, Wisconsin — offers all three zinc plating processes under one roof: barrel electroplating, rack electroplating, and mechanical galvanizing. ISO 9001:2015 certified, with compliance across ASTM B633, ASTM B695, and a full range of OEM specifications. On spec. On time. Or it is on them.
To request a quote or discuss your specific zinc plating requirements, contact Plateco at plateco.net or call (608) 524-8241.
This article is intended for informational purposes. Always consult with a qualified zinc plating specialist and reference applicable engineering specifications for your specific application requirements.