What Is Zinc Rack Electroplating
Zinc rack electroplating is a highly controlled electrochemical process in which individual steel or iron components are mounted on specialized racks, immersed in a zinc-ion solution, and subjected to an electrical current that bonds a precise zinc coating directly to the metal surface — creating the sacrificial galvanic protection that shields the underlying steel from corrosion.
The process is called “rack plating” because of its defining characteristic: unlike barrel electroplating — where parts tumble together in rotating drums — rack plating fixes each component individually onto purpose-built plating racks before it enters the electroplating bath. This individual treatment is what makes rack plating the method of choice for components that demand precision, consistency, and surface quality that bulk tumbling methods cannot deliver.
At Plateco, Inc. — Wisconsin’s ultimate zinc plating company — zinc rack electroplating is performed using advanced process control, rigorously maintained bath chemistry, and finishing expertise developed over decades of serving automotive suppliers, agricultural equipment manufacturers, industrial machinery producers, and OEM component makers across Wisconsin and beyond. The result is a consistent, on-spec zinc coating that can be engineered to meet the most exacting corrosion resistance standards in commercial manufacturing.
🔬 The galvanic protection principle: Zinc is electrochemically less noble than steel. When zinc coating is applied to steel, zinc becomes the sacrificial anode — it corrodes preferentially, protecting the underlying steel even when the coating is scratched or damaged. This self-healing corrosion protection mechanism is why zinc electroplating has been the dominant corrosion protection method in industrial manufacturing for over a century.
Understanding the complete rack electroplating process — not just the plating step itself, but every stage from initial cleaning through final quality inspection — is essential for engineers specifying zinc plating requirements, procurement managers evaluating plating suppliers, and manufacturers making decisions about corrosion protection for critical steel components. Every stage matters. Every variable affects the final coating quality. And every step that a plating provider shortcuts is a step that costs the customer in reliability, service life, and compliance.
// WHY_RACK_PLATING
Why Rack Plating — Not Barrel?
Before examining the process steps in detail, it’s worth understanding why rack plating is specified in the first place. Barrel electroplating and rack electroplating both deposit zinc from the same electrochemical process — but they serve fundamentally different component profiles and deliver different results.
| Parameter | Rack Electroplating | Barrel Electroplating |
|---|---|---|
| Part Size | Large, complex, heavy | Small, simple parts only |
| Coating Thickness Control | Precise to engineering spec | Less controllable — variable |
| Surface Finish Quality | Smooth, bright, cosmetically superior | Acceptable — less consistent |
| Complex Geometry Coverage | Excellent — custom rack fixtures | Limited on recesses & features |
| Part-to-Part Contact Damage | Zero — individual mounting | Possible — tumbling contact |
| Dimensional Tolerance Preservation | Superior | Less controlled |
| Cosmetic Quality | Ideal for visible components | Secondary to volume efficiency |
| Cost Efficiency for Volume | Higher per-part cost | More efficient for small parts |
| OEM Spec Compliance | Preferred for critical assemblies | Standard for commodity hardware |
The decision to specify rack plating is driven by the part itself. Components that are larger than what a barrel can process, that have complex geometries requiring coverage in deep recesses, that must meet strict dimensional tolerances after plating, or that need a cosmetically superior surface finish — all of these require rack plating. Plateco’s rack system at its Reedsburg, Wisconsin facility is capable of handling large, heavy components with customized rack fixtures engineered to match specific part geometries, ensuring consistent electrical contact and uniform current distribution across even the most complex shapes.
// PROCESS_OVERVIEW
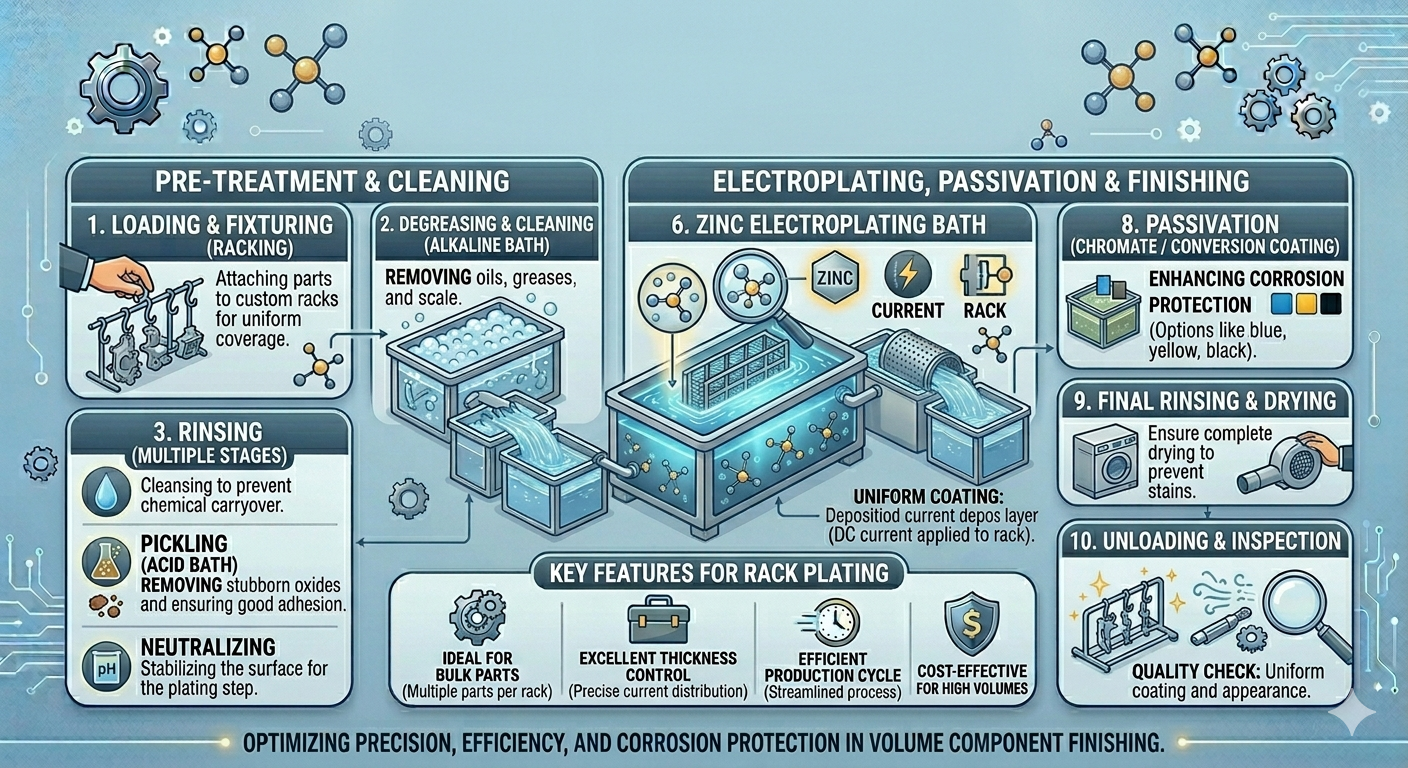
Process Overview: The Nine Stages
The complete zinc rack electroplating process at Plateco consists of nine interconnected stages, each of which must be executed correctly for the final coating to meet specification. There are no optional stages. A failure at any point in the sequence produces a defective coating — which is why Plateco’s approach treats zinc plating as an extremely complex process demanding state-of-the-art technology, painstaking planning, obsessive quality control, and a tremendous amount of talent.
⚠️ The shortcut problem: Many zinc plating companies treat the process as a simple dip-and-done operation — minimizing cleaning time, skipping rinse stages, or cutting corners on passivate application. Every shortcut taken at any stage directly compromises the quality, consistency, and corrosion resistance of the final coating. At Plateco, no stage is optional and no shortcut is acceptable.
The nine stages are: Surface Cleaning → Acid Activation → Rinse Cascade → Rack Mounting → Zinc Electrodeposition → Chromate Passivation → Sealing / Topcoat → Hydrogen Embrittlement Relief (when required) → Final Inspection & Quality Control.
Each stage is examined in detail in the sections that follow — with the technical rationale for every process parameter explained, so that engineers and procurement managers understand not just what Plateco does, but why each step is essential to delivering on-spec zinc rack plated components.
// PROCESS_STEPS
Steps 1 & 2: Surface Preparation & Cleaning
Surface preparation is the single most important stage in the entire zinc rack electroplating process. This is not an overstatement — it is an engineering reality confirmed by decades of plating practice. The zinc coating can only bond to the steel substrate as well as the substrate’s surface condition allows. Any contamination remaining on the part surface when it enters the plating bath will cause adhesion failure, blistering, incomplete coverage, or coating delamination in service.
Steel components arriving at a plating facility carry a predictable range of surface contaminants accumulated during manufacturing: cutting oils, forming lubricants, heat treat scale, mill scale, rust, weld flux, drawing compounds, and oxide layers. Every one of these must be completely removed before electroplating can begin. The cleaning sequence is deliberately multi-stage because no single chemistry removes all contaminant types effectively.
Alkaline Soak & Electrocleaning
Parts are submerged in heated alkaline cleaning solutions — typically caustic soda or proprietary alkaline cleaners — designed to saponify and emulsify oils, greases, and drawing compounds. Electrocleaning applies an electrical current to the part in the alkaline solution, generating gas bubbles that mechanically scrub the surface while the chemistry dissolves contaminants.
- Removes oils, lubricants, and forming compounds
- Heat acceleration improves cleaning efficiency
- Electrocleaning provides mechanical scrubbing via gas evolution
- Critical for parts with complex recesses where spray cleaning is ineffective
⚠️ Weld flux and heat treat scale are among the most difficult contaminants to remove. These chemically bonded surface layers require specialized cleaning chemistry and extended contact time — a detail that separates capable plating facilities from those who simply dip parts and proceed.
Multi-Stage Rinse Cascade
After each cleaning stage, parts are rinsed through a cascade of freshwater rinse tanks designed to remove cleaning chemistry and dissolved contaminants from the surface before the next stage. Rinse quality directly affects the performance of every subsequent stage — residual cleaning chemistry in the activation tank or plating bath contaminates those solutions and compromises coating quality.
- Multiple sequential rinse stages — not a single spray rinse
- Countercurrent design keeps early rinse stages cleaner
- Removes alkaline chemistry before acid stages prevent neutralization
- Water conductivity monitoring ensures rinse effectiveness
Step 3: Rack Mounting
Individual Part Fixturing on Plating Racks
Each part is carefully mounted onto specialized plating racks before entering the plating bath. Rack mounting is far more than a material handling step — it is an engineering exercise that directly determines coating uniformity, thickness consistency, and cosmetic finish quality. At Plateco, individual fixtures and hooks are configured to match each part’s specific geometry, providing optimized hook placement, stable positioning in the bath, and repeatable high-volume results.
- Ensures consistent electrical contact at each mount point
- Controls part orientation for optimal current distribution
- Prevents part-to-part contact that would mask surface areas
- Custom rack fixtures engineered for unique part geometries
- Stable positioning prevents movement that creates thickness variation
✅ The rack engineering advantage: At Plateco, racks are custom-engineered to match specific part profiles. This means that a complex bracket with deep pockets and irregular geometry receives the same current density and solution access on every surface — producing the uniform coverage that barrel plating can never achieve on complex parts.
Step 4: Acid Activation & Pre-Treatment Rinse
Acid Pickling & Surface Activation
Even after thorough alkaline cleaning, a thin oxide layer may persist on the steel surface — particularly if any delay occurred between cleaning and plating, or if the part was previously heat treated. Acid pickling immerses the part in a dilute acid solution (typically hydrochloric or sulfuric acid) that dissolves remaining oxide layers and etches the steel surface at the microscopic level. This etching creates a clean, reactive surface condition that allows the zinc layer to bond with maximum adhesion strength.
- Removes residual oxide and rust that alkaline cleaning cannot dissolve
- Activates the steel surface for optimal zinc nucleation
- Acid concentration, temperature, and contact time are precisely controlled
- Over-pickling (excess time or acid strength) can cause hydrogen absorption — monitored carefully
⚠️ Hydrogen absorption risk: Acid pickling causes hydrogen to be absorbed into the steel — a particular concern for high-strength steels (40+ HRC). This absorbed hydrogen can cause brittle fracture under load — called hydrogen embrittlement. Pickling time and acid strength are carefully controlled to minimize this risk, and high-strength parts receive a hydrogen embrittlement relief bake after plating (Step 8).
After acid activation, parts pass through a final rinse cascade that removes all acid residue before entering the plating bath. This rinse must be thorough — any carried-over acid contaminates the zinc bath chemistry and disrupts the plating process.
Step 5: Zinc Electrodeposition — The Core Process
Electrochemical Zinc Deposition
With the part surface perfectly clean and activated, the racks are immersed in the zinc electroplating bath — an aqueous solution containing zinc salts (typically zinc chloride or zinc sulfate, depending on bath chemistry type) along with carefully balanced additives that control brightness, ductility, and coating distribution. An electrical current is applied, causing zinc ions in the solution to migrate to the negatively charged parts (cathodes) and bond to the surface — atom by atom — building up the zinc coating layer.
- Current density precisely controls coating deposition rate
- Immersion time determines final coating thickness
- Bath chemistry (pH, zinc concentration, additive levels) is continuously monitored and adjusted
- Temperature regulation maintains consistent plating efficiency
- Rack orientation and anode placement ensure uniform current distribution
- Automated monitoring systems support repeatability across production batches
✅ Thickness engineering: Unlike barrel plating, rack plating allows engineers to dial in coating thickness to a specific target — adjusting current density and immersion time to achieve the precise deposit required by the engineering specification, whether that is a thin cosmetic coating or a heavy corrosion-resistance build-up meeting ASTM B633 requirements.
Bath Chemistry: Acid Zinc vs. Alkaline Zinc
The zinc plating bath type determines the coating’s character and the process’s capabilities. Acid zinc chloride baths offer faster deposition rates and excellent brightness — preferred for rack plating applications where cosmetic appearance matters. Alkaline zinc baths provide better throwing power (more uniform coverage into recesses and on complex geometries) and produce coatings with slightly better ductility. The choice between bath types is made based on part geometry, specification requirements, and final finish objectives.
Step 6: Chromate Passivation (Passivate Layer)
Trivalent Chromate Conversion Coating
After zinc deposition, the bare zinc surface is chemically reactive and begins to oxidize immediately when exposed to air and moisture — producing the white powdery zinc oxide and zinc hydroxide compounds that signal early zinc corrosion (“white corrosion”). Left unprotected, white corrosion can appear on zinc surfaces within days or even hours of exposure to humidity. The passivate step applies a chromate conversion coating that dramatically extends the zinc’s resistance to white corrosion and improves the coating system’s total corrosion protection performance.
- Passivate reacts chemically with the zinc surface to form a protective conversion film
- All Plateco passivates aretrivalent chromate— RoHS and REACH compliant, free of hexavalent chromium
- Passivate selection determines appearance (clear, yellow, black) and corrosion resistance level
- Contact time, solution concentration, and temperature are controlled for consistent film thickness
- The passivate film is self-healing at a microscopic level — providing dynamic protection
⚠️ Environmental compliance: Plateco uses exclusively trivalent chromate passivates — never hexavalent chromium compounds (Cr6+), which are classified carcinogens and banned or heavily regulated in most markets. Trivalent passivates deliver equivalent or superior corrosion protection while fully meeting RoHS, REACH, and automotive environmental directives.
Step 7: Sealing & Optional Topcoat
Wax or Polymer Sealer Topcoat
For applications demanding maximum corrosion protection or meeting the most stringent salt spray hour requirements, an additional topcoat sealer is applied over the passivate layer. Sealers — typically thin wax or polymer films — protect the passivate conversion coating from early mechanical abrasion and chemical attack, extending its effective life and thereby extending the total corrosion protection delivered by the complete coating system.
- Protects the passivate layer from damage during handling and assembly
- Significantly increases salt spray test performance hours
- Polymer sealers can also provide enhanced lubricity for assembly applications
- Applied via dip or spray — controlled film thickness for consistent performance
- Required for some OEM specifications — John Deere, Case CNH, Parker Hannifin, Caterpillar
✅ Three-layer system: The complete coating system — Zinc + Passivate + Sealer — is an engineered multi-layer defense. Each layer addresses a different failure mechanism: zinc provides sacrificial galvanic protection, passivate controls white corrosion onset, and sealer protects the passivate from mechanical damage. The combination can achieve hundreds of salt spray hours when properly applied.
Step 8: Hydrogen Embrittlement Relief Bake
Controlled Oven Bake for High-Strength Steels
High-strength steel components — typically those with hardness exceeding 40 HRC, including hardened fasteners, high-tensile structural components, and some spring steels — are susceptible to hydrogen embrittlement. During both acid pickling and electroplating, atomic hydrogen is generated at the steel surface and can be absorbed into the metal lattice structure. This absorbed hydrogen causes a phenomenon called delayed brittle fracture: the part may appear normal immediately after plating, but fail catastrophically under service load as the absorbed hydrogen causes internal micro-cracking.
- Required for all parts with hardness ≥ 40 HRC per ASTM B633 and most OEM specifications
- Controlled oven bake at temperatures between 375°F and 450°F (190–232°C)
- Bake duration — typically 3–24 hours — drives absorbed hydrogen out of the steel lattice
- Must be performed within 4 hours of electroplating for maximum effectiveness
- Elevated temperatures degrade the passivate film —parts must be re-passivated after baking
- Re-passivation restores full corrosion resistance before final inspection
⚠️ Re-passivation requirement: The oven bake temperature required for effective hydrogen relief destroys the chromate passivate film applied in Step 6. Parts must therefore return through the passivation line after baking to receive a fresh passivate coating — and for specifications requiring a sealer, the sealer must also be re-applied. This adds process time but is non-negotiable for high-strength steel safety.
Step 9: Final Inspection & Quality Control
Comprehensive Quality Verification
Because parts are plated individually on racks, the final inspection process at Plateco is more thorough and meaningful than what is possible after barrel plating — where the volume of parts processed together makes comprehensive individual inspection impractical. At Plateco, every batch undergoes systematic quality verification before shipment, ensuring that the on-spec commitment made to every customer is backed by documented evidence.
- Visual inspection— surface defects, coverage gaps, adhesion issues, cosmetic assessment
- Coating thickness measurement— X-ray fluorescence (XRF) or magnetic induction gauging to verify thickness meets specification
- Adhesion testing— standardized cross-hatch or bend tests confirm zinc-to-steel bond integrity
- Corrosion resistance evaluation— salt spray testing per ASTM B117 for specification compliance verification
- Certificate of conformance— documentation confirming specification compliance for customer quality systems
- Dimensional verification— confirming coating thickness did not affect critical dimensional tolerances
✅ Plateco’s quality commitment: In Q1 2026, Plateco’s customer return rate was 0.1%. This extraordinary figure — achieved for the 6th consecutive year as a recognized Top Shop — reflects what happens when every stage of the plating process is treated with equal seriousness: defects become the exception, not the baseline.
Finish Options, Quality Pillars & FAQs
Available Finish Options
The passivate and sealer selections applied in Steps 6 and 7 determine the final appearance and corrosion resistance performance level of the finished part. Plateco offers five finish options engineered to meet diverse application requirements:
Clear Zinc
Classic bright appearance. Solid white corrosion protection. Industry standard for most industrial applications.
Yellow Chromate
Iridescent gold appearance. Enhanced corrosion resistance vs. clear. Widely specified for demanding environments.
Black Trivalent
Dark, aesthetically distinctive finish with functional corrosion protection. RoHS & REACH compliant.
Olive Drab
Military and defense industrial applications. Specific corrosion and appearance performance targets.
Custom Finishes
Engineered to OEM specification — John Deere, Case CNH, Caterpillar, Parker Hannifin, and others.
Quality Pillars at Plateco
🎯 On Spec. Always.
If Plateco delivers a plated part that you reject for a quality problem that is their fault, you will not be charged for that part. This is an unconditional quality commitment — not a marketing claim.
⏱️ On Time. 95% of the Time.
Plateco delivers on time 95% of the time — a verifiable metric that eliminates the missed delivery excuses that plague manufacturers relying on less disciplined plating suppliers.
📋 ISO 9001:2015 Certified
Plateco’s quality management system is certified to ISO 9001:2015 — providing the systematic process documentation, audit trails, and continual improvement framework that OEM suppliers require.
🏆 Top Shops — 6 Consecutive Years
Recognized as a Top Shop for the 6th straight year — an industry award reflecting measurable excellence in quality systems, delivery performance, and customer satisfaction.
Frequently Asked Questions
Ready for Rack Plating Perfection?
Tell Plateco about your part specifications, volume, engineering requirements, and corrosion targets — and the team will recommend the exact rack electroplating solution your application demands.
// 1375 Industrial Street, Reedsburg, WI 53959 | (608) 524-8241 | ISO 9001:2015
// continue reading